Promepla OEM Solutions has 20 years experience in Manufacturing, Assembly and Packaging of Single Use Medical Devices. Promepla has experience in assembly of complex devices for the pharmaceutical, drug delivery, medical and healthcare industries. Cleanroom Facilities include Two (ISO class 8 / US class 100 000) cleanrooms over 2 000 sq meters for assembly and packaging. One (ISO class 8 / US class 100 000) cleanrooms over 200 sq meters for injection. Cleanrooms certified to ISO 14644 for airborne particulate Possibility to work under a more restrictive environnement if requested or necessary. (ISO7 or less) Micro organism monitoring (on product, surfaces and air) ensures environment compliance and future sterility of the product. Our manufacturing capabilities include:
CLEANROOM MANUFACTURING & ASSEMBLY
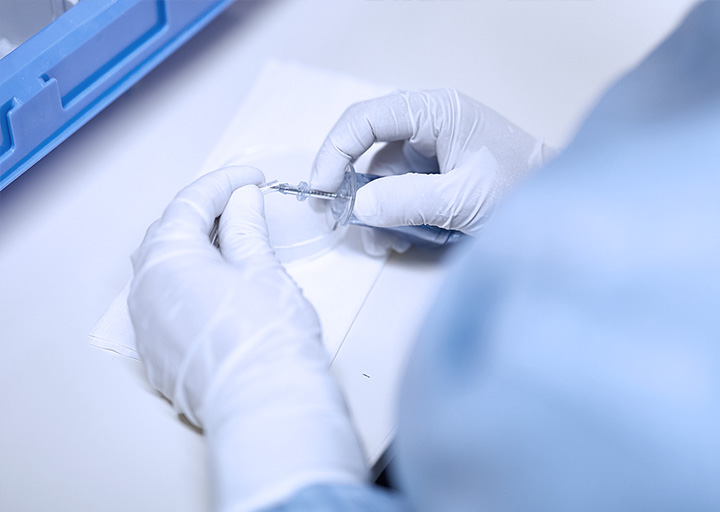
CLEANROOM MANUFACTURING & ASSEMBLY